When the solid phase (ice) changes to a liquid, melting occurs and heat is also absorbed. 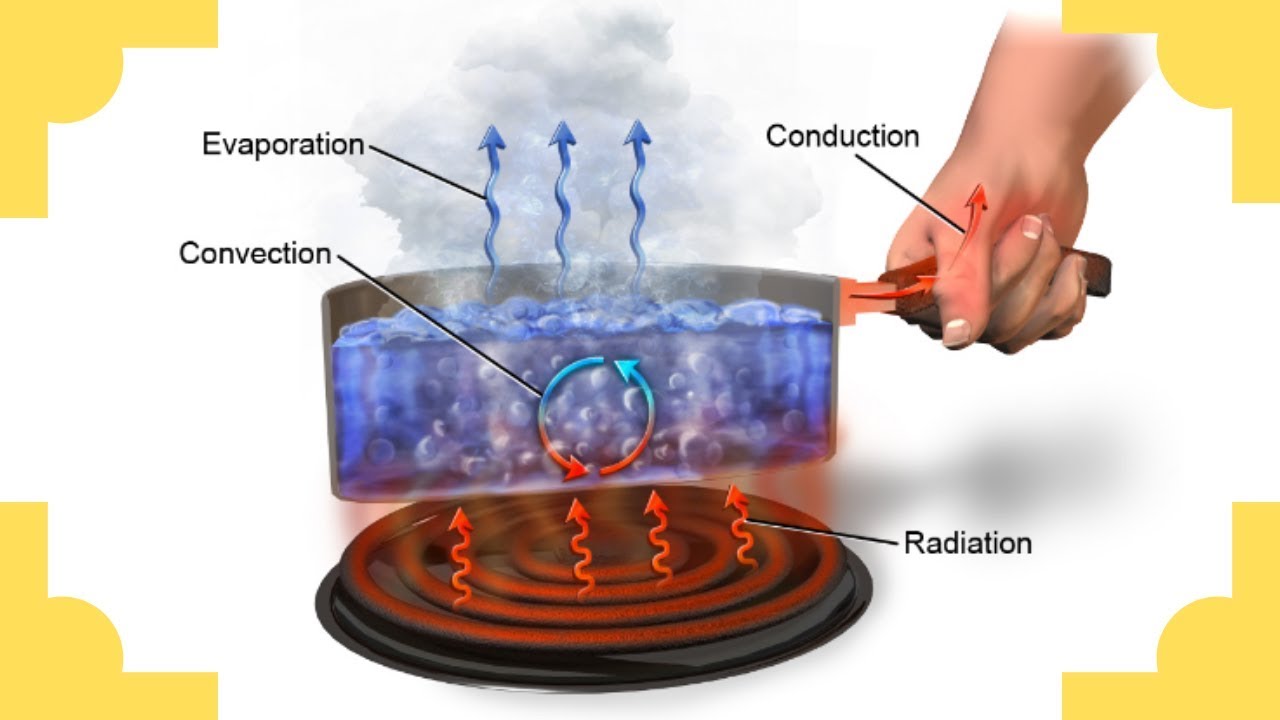
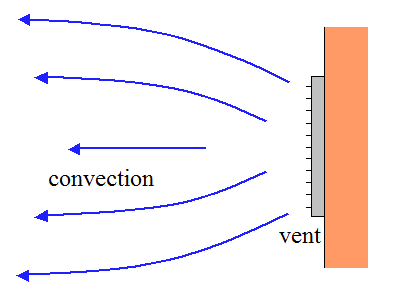
This process is known as evaporation, and the absorption of heat is called the latent heat of evaporation (or latent heat of vaporization). This energy that is absorbed gives the molecules the extra motion that is needed to escape the surface of the liquid to become a gas. Heat is absorbed when water changes from a liquid to a gas (water vapor). This type of heat is called latent heat, because that heat is stored or hidden until the phase change occurs. When water changes phase, heat is exchanged between the water and its surroundings - the water either absorbs or releases heat depending on the phase change. Extra energy is needed to break this strong bond and change water from one phase to another. The reason that heat is transferred as water changes phase is due to the hydrogen bond between molecules of water. In other words, it changes from a liquid, solid, or gas (water vapor) into a different form or phase (melting ice is an example of a phase change). We can sense the resulting change in temperature, so these heat transfer mechanisms are known as sensible heating.Īnother type of important heat transfer process affecting the climate system occurs when water undergoes a change in phase. As air or water cools, its density increases and it sinks.Ĭonduction and convection work together to transfer heat. 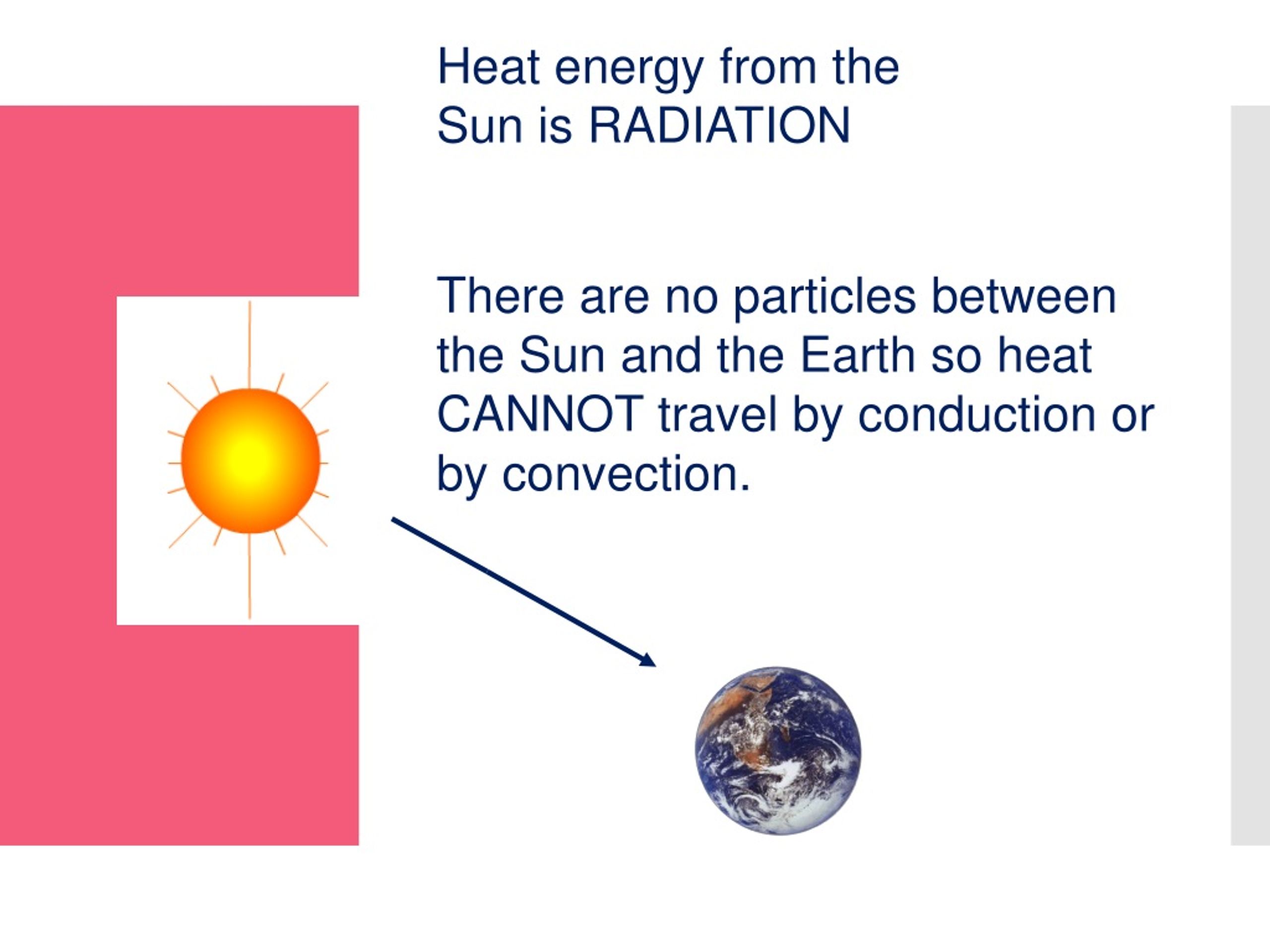
As air or water warms, it expands and becomes less dense than the air or water above, and it rises. Convection currents form when there is unequal heating of the atmosphere or water. Consequently, convection can occur in the atmosphere or in bodies of water. Convection is the spread of heat in a fluid, defined as a gas or liquid in which atoms and molecules are moving relatively freely. Heat is transferred vertically in the troposphere by convection. The W refers to watt, which is the unit used to express power (expressed in joules per second). The Greek letter “ σ” (sigma) represents the Stefan-Boltzmann constant (5.67 x 10 -8W/m 2K 4) and T is the object's surface temperature in Kelvin. E = σT4 Stefan-Boltzmann LawĮ represents the maximum rate of radiation (often referred to as energy flux) emitted by each square meter of the object's surface. This law (expressed mathematically as E = σT4) states that all objects with temperatures above absolute zero (0K or -273☌ or -459☏) emit radiation at a rate proportional to the fourth power of their absolute temperature. The Stefan-Boltzmann law, a fundamental law of physics, explains the relationship between an object's temperature and the amount of radiation that it emits. For this reason, the following two laws (Stefan-Boltzmann and Wein's laws) can be used to explain the correlation between temperature and radiation for the sun and Earth. An object that absorbs and emits all possible radiation at 100 percent efficiency is called a blackbody. Absolute zero is equal to zero Kelvin, which is equal to -273☌ or -460☏.īoth the sun and Earth's surface behave as blackbodies. All objects actually emit radiation if their temperature is greater than absolute zero. For example, when a stovetop burner reaches 1,000 Kelvin (K) - 726° Celsius (C) or 1,340° Fahrenheit (F) - it will glow red. When an object is hot enough, you can see the radiation it emits as visible light.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
